 The American Congress of Obstetricians & Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) have issued an updated joint statement on noninvasive prenatal genetic screening (NIPGS). It adds clarity on whom practitioners should offer it to, what NIPGS tests for, and how it relates to other prenatal testing options.
The American Congress of Obstetricians & Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) have issued an updated joint statement on noninvasive prenatal genetic screening (NIPGS). It adds clarity on whom practitioners should offer it to, what NIPGS tests for, and how it relates to other prenatal testing options.
2012 ACOG/SMFM Statement
NIPGS entered the commercial market in October 2011 when Sequenom launched MaterniT21. A year later, ACOG and SMFM issued the joint statement that guided practitioners on how NIPGS should be used and guided insurers on when it should be covered.
The significant limitation ACOG/SMFM placed on NIPGS was recommending it only be offered to women considered “high risk.” These patients had at least one of the following characteristics:
- 35 years of age or older;
- Had a prenatal screening finding indicative of increased chance for having a child with aneuploidy;
- Had had a previous pregnancy positive for aneuploidy; or,
- One of the parents had a balanced translocation.
While this population has risen in the past years as more women are having more children later in life, by definition, most women are not “high risk.” The reason for the limitation was that at the time of the 2012 statement, published studies were limited to women who were considered high risk.
General population studies
In the ensuing years, studies began being published about NIPGS’ performance in the general population, i.e. all expectant women, not just those considered high risk.
In 2014, a study with researchers affiliated with Illumina, the maker of verifi, published a study in the New England Journal of Medicine (NEJM) that reported NIPGS in the general population had a 45.5% positive predictive value. For this, and other limitations in the study, the SMFM issued a statement reminding practitioners that its recommendations remained to limit the offering of NIPGS to high risk mothers.
In 2015, a study with researchers affiliated with Ariosa, the maker of Harmony, also published a study in the NEJM of Harmony’s performance in the general population. This time, ACOG issued a practice advisory noting the developments of NIPGS, saying ACOG was paying attention to them, but that its recommendations on NIPGS had not been changed … yet.
2015 ACOG/SMFM statement on NIPGS
With the increasing number of NIPGS published studies, ACOG and SMFM have issued an updated statement. Notably, it no longer limits the offering of NIPGS to high risk mothers. But, tellingly, it does not recommend offering NIPGS to most women. It further clarifies what conditions NIPGS is recognized as a valid screen for and how it should be integrated with other prenatal testing options.
Who
ACOG/SMFM recognizes that
Although any patient may choose cell-free DNA analysis as a screening strategy for common aneuploidies regardless of her risk status, the patient choosing this testing should understand the limitations and benefits of this screening paradigm in the context of alternative screening and diagnostic options.
That said, because NIPGS has a lower positive predictive value in the low-risk population, ACOG/SMFM say that:
Given the performance of conventional screening methods and the limitations of cell-free DNA, conventional screening methods remain the most appropriate choice for first-line screening for most women in the general obstetric population.
Therefore, ACOG/SMFM have removed the limitation of offering NIPGS to only high risk women. But, as most women are not high-risk, conventional screening methods, i.e. the nuchal-translucency combined test, the quad test, and combinations thereof, are the most appropriate screens for most women.
The 2015 statement does, though, impose a limitation for some expectant mothers: those expecting multiples. Because the published studies have not shown the same level of accuracy in pregnancies with twins, triplets, etc. as singleton pregnancies, the 2015 statement does not recommend NIPGS for women with multiple gestations.
What
Soon after MaterniT21 launched, Illumina entered the market with an expanded panel of conditions including sex aneuploidies. Not to be outdone, Sequenom rebranded its test as MaterniT21 Plus to emphasize it tests for Down syndrome, plus trisomy 18 and 13 and sex aneuploides. Then, with each passing year, laboratories promoted their expansion into other conditions like triploidy (Natera) and microdeletions (Sequenom, Illumina, and Natera). In 2015, it was announced that NIPGS also could report findings indicating cancer in the mother.
The ACOG/SMFM 2015 statement, however, makes clear that there is no “plus.” It recognizes NIPGS as a screen for four conditions: trisomies 21, 18, and 13 and y-based testing to indicate the sex of the fetus. From the statement:
The cell-free DNA test will screen for only the common trisomies and, if requested, sex chromosome composition.
The statement includes another bullet point on the limited scope of conditions NIPGS is recognized as screening for:
Routine cell-free DNA screening for microdeletion syndromes should not be performed.
How
The 2015 statement provides instruction on how NIPGS should be administered in clinical practice.
The alpha and omega of the bulleted highlights of the statement is to advise practitioners that women should receive pre-test counseling on the available prenatal testing options and their respective limitations, and that practitioners should respect that:
Patients may decline all screening or diagnostic testing for aneuploidy.
For those women who do accept NIPGS, the statement addresses how results should be delivered and what follow-on testing, if any should be recommended:
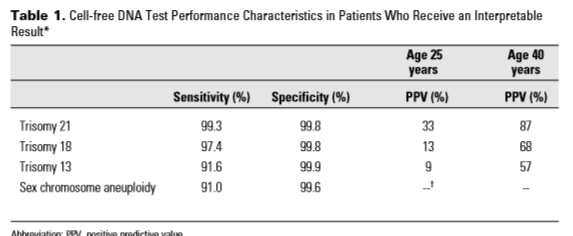
- While the statement notes laboratories currently report results as “positive” or “negative” or “indicative of high risk,”these reporting methods are not as useful for providers as reporting the positive predictive value (PPV) and residual risk. The statement includes a table demonstrating how a “positive” screen result could vary widely based on the mother’s age for reporting the actual chance she may have an affected pregnancy:
ACOG/SMFM encourage laboratories to report findings with PPV and residual risk.
- If the mother receives a “no call,” meaning the laboratory could not report a result based on the sample tested, then the mother should receive comprehensive ultrasound evaluation and be offered diagnostic testing due to increased chance for aneuploidy; in published studies, the patients receiving “no calls” had a high percentage of actually having an affected pregnancy.
- If the mother receives a “negative,” she should be counseled on the chance of it being a “false negative” and that she may still have an affected pregnancy; the statement notes a study that found women who had a conventional screen indicative of aneuploidy but who received a negative NIPGS, ended up having a 2% chance of having an affected pregnancy.
The statement further advises on how NIPGS should be used with traditional screening. While the statement says other screening methods for aneuploidy should not be used in parallel or simultaneously with NIPGS, women should be offered screening for structural defects, like open neural tube defect, since NIPGS does not screen for these conditions.
Finally, the report reiterates the 2012 statement’s emphasis on NIPGS being a screen and therefore diagnostic testing should be offered to confirm a positive result. Specifically:
Management decisions, including termination of the pregnancy, should not be based on the results of the cell-free DNA screening alone.
More information
The ACOG/SMFM 2015 statement authorizes what some practitioners were already doing: offering NIPGS to all women. However, it does not recommend this practice. Rather, it recognizes women may choose to have NIPGS, but for most women, i.e. low risk women, conventional screening remains the recommended first-tier option. Should patients have NIPGS, it is more useful for laboratories to report the PPV, and confirmation of a screen-positive should be made by diagnostic testing, particularly prior to any decision to terminate.
The ACOG/SMFM 2015 statement ends with a link to additional information and resources that ACOG/SMFM notes will be helpful for practitioners. In addition to sharing a link to the statement itself, it shares links to fact sheets produced in 2015 by the National Society for Genetic Counselors for both practitioners and patients. These fact sheets cover frequently asked questions about NIPGS and its results, and share recommended resources for patients.

4 thoughts on “Noninvasive prenatal genetic testing not recommended as first tier screen for most women”